Legislative Mandate: Section 22C(1)(b) of MEDICINES AND RELATED SUBSTANCES ACT 101 OF 1965 as amended and Regulation 23.
The Pharma Licencing sub-unit is responsible for issuing to a manufacturer, wholesaler or distributor of a medicine or scheduled substance, a licence to manufacture, import, export, act as a wholesaler of or distribute, as the case may be, such medicine and/or scheduled substance, upon such conditions as to the application of such acceptable quality assurance principles and good manufacturing and/or distribution practices as the Authority may determine.
For facilities handling Medical Devices or Complementary Medicines, refer to the appropriate webpages.
Pharma Licenced Establishments
Pharma Licencing Guidelines
Pharma Licencing Forms
Submission for the following licence applications only:
Application Supporting Documents
Completed application forms with the following supporting documents:
- Licence application cover Letter
- Proof of payment with the correct reference as per Annexure A – SAHPRA Fee Categorisation Guideline
- Latest Inspection resolution if applicable
- Existing Licence being renewed/amended if applicable
- CIPC/CIPRO/DTI CERTIFICATES OR DOCUMENTS proving ownership of the business
- NDOH PREMISES LICENCE (OR COPY OF COMPLETED APPLICATION FORM – evidence of having applied)
- REGISTRATION AS RESPONSIBLE PHARMACIST (OR SAPC CASE NUMBER)
- SAPC CERTIFICATE OF RECORDING OF A PHARMACY (OR SAPC CASE NUMBER)
- SAPC CERTIFICATE OF RECORDING OF A PHARMACY OWNER (OR SAPC CASE NUMBER)
- Copy of POP of the licence annual retention fee for all preceding years if licence is older than a year.
- Product List with POP of annual retention fee for each product since the product was registered.
Applicants to submit a zipped file.
Fees information below:
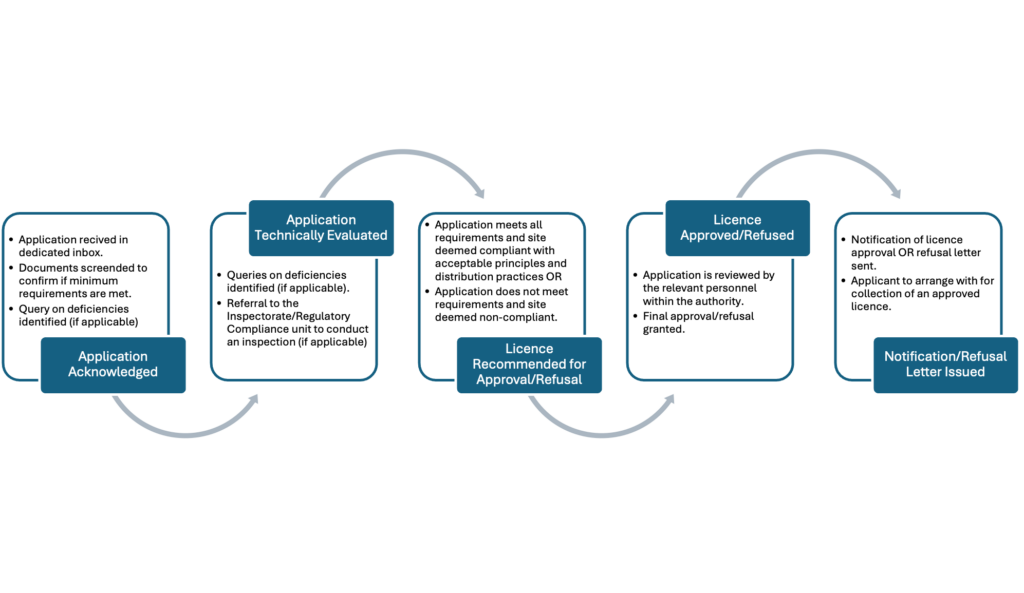
Application Process Flow